Lipid nanoparticles (LNPs) are an integral part of the mRNA vaccine technology. They are being used to deliver mRNA vaccines into your cells. But are they immunologically inert?
Lipids are not meant to be injected. When injected, they induce inflammation. The immune system registers ‘foreign’ lipids as a ‘danger’ signal because they indicate (or mimic) the presence of pathogenic invaders, such as bacteria. A bacterial component alone can activate the immune system via pathogen-associated molecular pattern (PAMP) receptors, even when an actual infection is not present.
In response to ‘danger’ signals, the immune system starts producing pro-inflammatory cytokines, which leads to body aches, fever, chills, malaise, etc. And if the production of these cytokines is excessive or unmitigated by anti-inflammatory safeguards, a ‘cytokine storm’ (formally known as Cytokine Release Syndrome, or CRS) may ensue with intolerable or even deadly outcomes.
In the 1940s, immunologist Jules Freund discovered that mineral oil can be used as an adjuvant (a ‘helper’) to activate the immune system. Freund’s oil-based adjuvants never reached licensure for human use due to their severe inflammatory side effects. Even in animal research, the use of these adjuvants is strictly regulated by ethical guidelines due to painful reactions they produce.
What types of lipids can potentially end up in mRNA vaccines?
In addition to lipid nanoparticles that are used for mRNA encapsulation, these vaccines might be contaminated with bacterial lipids. This is because the first step in the mRNA vaccine manufacturing process is growing a plasmid DNA template in E. coli cells, from which mRNA is then synthetically transcribed. E. coli are Gram-negative bacteria that contain an endotoxin called lipopolysaccharide (LPS), which is a potent inflammatory substance that leads to the immune system activation.
While drugs are required to be rigorously tested for pyrogenic (fever-inducing) substance contamination, such tests are not required for biologics, including vaccines, per Code of Federal Regulations CFR 610.13. The amount of LPS contamination in mRNA vaccines is therefore unknown and may vary from batch to batch. And despite high rates of inflammatory side effects observed in mRNA vaccine recipients, our regulatory health authorities have turned a blind eye to the lipid content in mRNA vaccine formulations.
What are the systemic side effects observed in mRNA vaccine recipients?
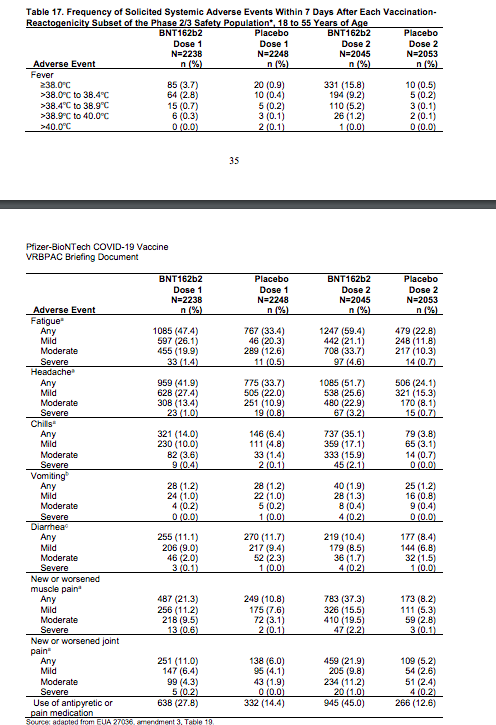
Systemic reactions documented in the Pfizer vaccine trial recipients at very high rates include fever, fatigue, headaches, chills, muscle or joint pain, and even vomiting. The frequency and severity of these reactions increase alarmingly after the second vaccine dose, reaching in some instances over 50% (Table 17, p. 35-36).
December 10, 2020
https://www.fda.gov/media/144245/download
One of the vaccine trial participants described the difference in how she felt after the first versus the second dose. She published a formal account of her experience in the Journal of American Medical Association (JAMA).
After the first injection, she recalls: “My arm was sore, but I did not notice anything out of the ordinary. I could not begin to guess whether I had received the vaccine or the placebo.”
A totally different story after the second injection: “The experience after the second injection was different. My arm quickly became painful at the injection site, much more than the first time. By the end of the day, I felt light-headed, chilled, nauseous, and had a splitting headache. I went to bed early and fell asleep immediately. Around midnight, I woke up feeling worse—feverish and chilled, nauseated, dizzy, and hardly able to lift my arm from muscle pain at the injection site. My temperature was 99.4 °F (37.4 °C). I tossed and turned, sleeping little during the rest of the night. When I woke up again at 5:30 am, I felt hot. Burning. I took my temperature and looked at the reading: 104.9 °F (40.5 °C). This was the highest fever I can ever remember having, and it scared me. I took acetaminophen and drank a glass of water. When the research office opened at 9 am, I called to report my reaction to the injection. Thankfully, my fever had come down to 102.0 °F (38.9 °C) by then. The research nurse said, “A lot of people have reactions after the second injection. Keep monitoring your symptoms and call us if anything changes.”
Why does the escalation of symptoms happen after the second dose?
Innate immunologic memory is at play here. Immunologic memory means an augmented response to the second exposure. For a long time, immunologic memory was thought to occur only within adaptive immunity, but this view has recently changed. The authors of the 2017 Review on Nanoparticles and Innate Immunity mention the following in section 3.3 Nanoparticles and Modulation of Innate Immune Memory: “The ability of innate immune system to develop an immunological memory is a recent re-discovery in immunology, a concept that goes against the dogma that only adaptive immunity can develop memory.”
While the adaptive immune system responds to antigens and produces antigen-specific antibodies, the innate immune system is triggered by inflammatory substances and produces cytokines. Due to innate immunologic memory, the second exposure to an inflammatory substance would result in the unwanted escalation in cytokine production and lead to higher frequency and severity of systemic inflammation, which is what would make you feel sicker after the second dose.
Then what do you gain after getting through these severe vaccine side effects, which can only get worse with each subsequent dose? Do you gain protection from COVID-19 for life? Not necessarily.
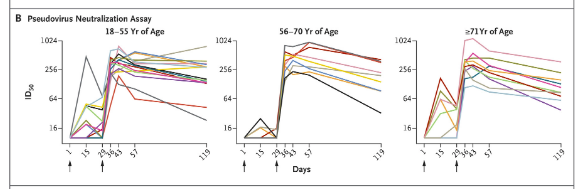
Neutralizing, as opposed to binding, antibodies are the ones that give you protection from the virus. Widge et al. (2021) measured neutralizing serum titers in the mRNA vaccine recipients after the first and second doses for 119 days (4 months) after vaccination. The titers went down (in some cases all the way to the background level!) within 4 weeks after the first dose, necessitating a booster at that time. After the booster, protective titers reached higher levels than the first time, in accordance with the adaptive immunologic memory. But then they too started waning noticeably. It is clear from their downward trend that they will not stay up for too long beyond the 119 days. The duration of vaccine protection after the 2-dose schedule is entirely uncertain at this point, as vaccine trials lasted for barely 2-3 months before the emergency authorization. Are you ready to be told that more boosters will be required in the future?
Moving forward, we need safer and more sustainable solutions than what mRNA vaccines can provide, such as strengthening the immune system to withstand COVID-19 itself and utilizing the available therapeutics that have demonstrated high rates of success in COVID-19 recovery. And the uncoerced choice to vaccinate or not should always be left up to individuals.

